The industrial chemistry sector faces unprecedented challenges in separating actinides from rare earths, two element families that share remarkably similar chemical properties. This separation difficulty creates significant bottlenecks across nuclear waste management and critical mineral supply chains. The challenge stems from fundamental atomic structure similarities that make these elements behave almost identically in most chemical environments, requiring sophisticated approaches that integrate advanced uranium market dynamics and emerging separation technologies.
Understanding the Chemical Challenge Behind Actinide-Lanthanide Separation
The core difficulty in separating actinides from rare earths lies in their nearly identical chemical behaviour patterns. Both element families predominantly exist in the +3 oxidation state under typical processing conditions. Consequently, this creates a situation where conventional separation techniques prove inadequate for achieving high purity requirements.
Why These Elements Behave Like Chemical Twins
Chemical similarity between actinides and lanthanides manifests in several critical ways. Both families form hydrated ions with comparable charge-to-radius ratios, resulting in similar hydration sphere structures. Furthermore, lanthanides typically form hydrated complexes [Ln(H₂O)ₙ]³⁺ with coordination numbers of 8-9, while actinides in the +3 state exhibit comparable hydration behaviour.
This similarity extends to complexation chemistry with common ligands including nitrates, chlorides, and phosphates. Distribution factors in single-stage extraction processes typically range from 1.5 to 5 for actinide-lanthanide separations, significantly lower than the 10-100 range achievable in favourable rare earth separations.
Key chemical similarities include:
• Overlapping hydrolysis equilibria at neutral to acidic pH ranges
• Comparable stability constants with halide and pseudohalide ligands
• Similar extraction efficiency in classical solvent systems
• Matching trends in complex formation with organic extractants
The Fundamental Chemistry That Makes Separation Difficult
The separation challenge intensifies due to overlapping ionic radii between the two families. Actinide ions in the +3 state exhibit ionic radii ranging approximately 1.00-1.10 Å, while lanthanide ions span 0.86-1.03 Å. However, this overlap creates conditions where size-selective separation mechanisms fail to provide adequate selectivity.
Unlike lanthanides, which remain predominantly in the +3 oxidation state under aqueous conditions, actinides can access multiple stable oxidation states. For instance, uranium commonly exists as +4 or +6 (uranyl UO₂²⁺), neptunium can adopt +4, +5, or +6 states, and plutonium exhibits +3, +4, and +6 oxidation chemistry.
Oxidation States and Ionic Radii Similarities
The oxidation state variability of actinides provides potential separation advantages not available with lanthanides. Selective oxidation or reduction can shift actinide distribution coefficients, creating separation opportunities. Nevertheless, this approach requires precise redox control and adds operational complexity to industrial processes.
Oxidation state considerations:
• Thorium: predominantly +4 oxidation state
• Uranium: +4 and +6 states under different conditions
• Americium: primarily +3, with +6 accessible under specific conditions
• Lanthanides: essentially limited to +3 under typical processing conditions
When big ASX news breaks, our subscribers know first
What Are the Primary Industrial Separation Technologies?
Industrial separation of actinides from rare earths relies primarily on three established technology platforms, each offering distinct advantages and operational characteristics. These methods have evolved from decades of research in both nuclear fuel processing and rare earth refining applications.
Liquid-Liquid Solvent Extraction Systems
Solvent extraction represents the dominant industrial approach for separating actinides from rare earths, accounting for approximately 95-98% of commercial separation capacity globally. This method operates through selective partitioning of metal species between aqueous and organic phases using specialised extractants.
The process requires multiple equilibrium stages configured in counter-current cascades to achieve high separation efficiency. Typically, industrial systems employ 5-15 stages for actinide-lanthanide separations, significantly more than the 2-4 stages often sufficient for lanthanide-lanthanide separations.
Critical operating parameters include:
• pH control within narrow ranges (typically 1.5-3.5)
• Temperature management to optimise equilibrium selectivity
• Extractant concentration optimisation for maximum distribution ratios
• Phase ratio control to balance throughput and separation efficiency
Modern solvent extraction facilities incorporate automated pH adjustment systems and continuous monitoring to maintain optimal separation conditions. In addition, the organic phase typically contains specialised extractants dissolved in hydrocarbon diluents, while the aqueous phase composition varies based on feed material and target products.
Ion Exchange and Chromatographic Methods
Ion exchange separation utilises functionalised resins to selectively bind actinides or lanthanides based on differences in charge density and complexation preferences. While representing less than 5% of primary separation capacity globally, ion exchange offers advantages in specific applications including recycling operations and high-purity material production.
Modern resins incorporating diphosphonic acid ligands achieve selectivity coefficients (α) of 3-8 for americium-europium separations under optimised conditions. These systems require careful pH buffering and extended residence times but can produce ultra-high purity products with minimal chemical waste generation.
Ion exchange advantages:
• Reduced chemical footprint compared to solvent extraction
• Higher product purity potential
• Better suited for small-scale or specialised applications
• Lower organic solvent requirements
Emerging Electrochemical and Pyroprocessing Routes
Pyroprocessing technologies operate in molten salt environments at elevated temperatures (450-850°C), typically using LiCl-KCl eutectic mixtures as the electrolyte medium. These systems separate metals through electrochemical potential differences, with actinides typically depositing at more negative potentials than lanthanides.
Electrochemical separation offers particular advantages for processing materials with high radiation fields, as molten salt systems demonstrate superior radiation tolerance compared to organic solvent-based methods. Moreover, the high-temperature environment accelerates reaction kinetics, enabling faster processing cycles.
| Method | Efficiency Rate | Primary Application | Key Advantages | Limitations |
|---|---|---|---|---|
| Solvent Extraction | 90-95% | Industrial scale processing | Proven scalability | Complex chemistry control |
| Ion Exchange | 85-92% | High-purity applications | Minimal waste generation | Limited throughput |
| Pyroprocessing | 88-94% | High-radiation environments | Radiation tolerance | High temperature requirements |
| Hybrid Systems | 95-98% | Specialised applications | Combined benefits | Increased complexity |
How Do ALSEP and TALSPEAK Processes Work in Practice?
Advanced separation processes like ALSEP and TALSPEAK represent specialised approaches developed specifically for nuclear applications where traditional methods prove insufficient. These technologies incorporate sophisticated chemistry to achieve separation factors significantly higher than conventional solvent extraction.
ALSEP Process Design and Operating Parameters
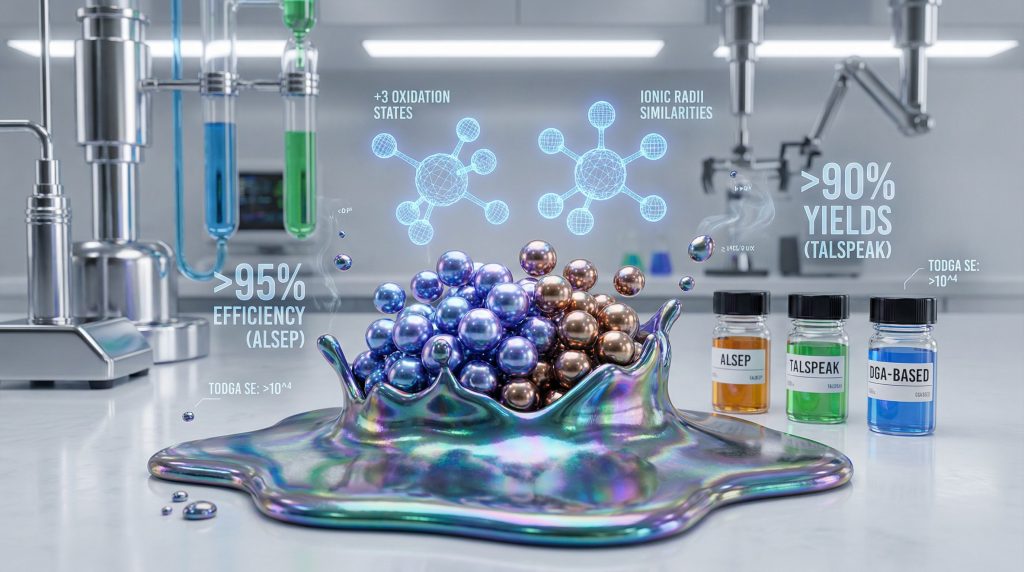
The ALSEP (Actinide Lanthanide SEParation) process operates through carefully controlled pH conditions to maximise selectivity between actinide and lanthanide species. Laboratory demonstrations have achieved separation efficiencies exceeding 95% under optimised conditions, though full-scale commercial deployment remains primarily at pilot scale outside national laboratory facilities.
ALSEP systems require precise pH control within the 2.0-3.5 range, where actinide and lanthanide complexation behaviour diverges sufficiently to enable effective separation. Furthermore, the process utilises specialised extractants that exhibit enhanced selectivity compared to traditional phosphorus-based reagents.
Critical ALSEP parameters:
• pH maintenance within ±0.1 units for optimal selectivity
• Temperature control between 20-40°C to balance kinetics and thermodynamics
• Multi-stage counter-current operation for high recovery
• Automated monitoring systems for process stability
TALSPEAK Method for High-Yield Separations
TALSPEAK (Trivalent Actinide Lanthanide SePAration by Phosphorus-reagent Extraction from Aqueous Komplexes) achieves separation through a bifunctional approach combining selective extractants with aqueous-phase complexants. The method was developed at Oak Ridge National Laboratory and demonstrates separation factors of 10-50 under optimised conditions.
The TALSPEAK mechanism relies on preferential lanthanide complexation with DTPA or EDTA in the aqueous phase while actinides partition into the organic phase containing CMPO extractant. This dual-action approach enables rapid equilibration, with typical contact times of 5-30 minutes per stage compared to hours required for conventional methods.
TALSPEAK operational specifications:
| Parameter | Optimal Range | Function |
|---|---|---|
| pH (aqueous) | 2.0-2.5 | Complexant stability |
| CMPO concentration | 0.01-0.1 M | Organic phase extractant |
| DTPA concentration | 0.05-0.2 M | Aqueous phase complexant |
| Contact time | 5-15 minutes | Equilibration period |
| Temperature | 20-40°C | Selectivity optimisation |
Diglycolamide-Based Extractant Systems
Diglycolamide (DGA) extractants represent an emerging class of separation reagents offering advantages over traditional phosphorus-based systems. These compounds, including TODGA and TEHDGA variants, operate through neutral solvation complex formation with metal nitrates rather than cation exchange mechanisms.
DGA-based systems demonstrate moderate selectivity (separation factors of 2-5 for americium-europium) but offer operational benefits including reduced radiolysis susceptibility and compatibility with multiple acid systems beyond nitric acid. Additionally, the neutral extraction mechanism simplifies aqueous phase chemistry and reduces secondary waste generation.
Key DGA advantages:
• Enhanced radiation stability compared to phosphorus extractants
• Compatibility with HCl and H₂SO₄ systems
• Simplified back-extraction chemistry
• Reduced organic phase degradation products
| Extractant Type | Selectivity Factor | Acid Compatibility | Commercial Status | Key Benefits |
|---|---|---|---|---|
| TODGA | 2-4 | HNO₃/HCl | Pilot demonstration | Proven chemistry |
| TEHDGA | 3-6 | Multiple acids | Research phase | Enhanced selectivity |
| DMDODGA | 2-5 | HCl optimised | Development stage | Reduced steric effects |
Which Industries Drive Demand for Actinide-Lanthanide Separation?
Industrial demand for effective actinide-lanthanide separation stems from three primary sectors, each with distinct technical requirements and economic drivers. These industries collectively represent billions of dollars in annual production value and face increasing pressure to achieve higher purity standards while managing regulatory compliance costs.
Nuclear Fuel Cycle and Waste Management Applications
The nuclear industry represents the most technically demanding application for actinide-lanthanide separation, where high radiation fields and strict purity requirements create unique processing challenges. Spent nuclear fuel contains both actinides and fission product lanthanides that must be separated for recycling, waste form optimisation, and transmutation applications.
Advanced nuclear fuel cycles require separation of minor actinides (americium, curium, neptunium) from lanthanide fission products to enable transmutation technologies that could significantly reduce long-term radioactive waste burdens. Current separation technologies achieve 95-99% efficiency under laboratory conditions, but scaling to industrial throughput while maintaining performance remains challenging.
Nuclear separation drivers:
• Advanced fuel cycle development programmes
• High-level waste volume reduction initiatives
• Transmutation reactor feedstock preparation
• Repository thermal load management requirements
Rare Earth Mining and Purification Requirements
Natural rare earth deposits frequently contain actinides, particularly thorium and uranium, requiring effective separation to produce market-acceptable products. Monazite processing, which accounts for a significant portion of global heavy rare earth production, must achieve thorium removal to parts-per-million levels for most commercial applications.
Regulatory frameworks in most developed countries impose strict limits on actinide contamination in rare earth products, particularly for applications in consumer electronics and automotive components. Consequently, these requirements drive demand for separation technologies capable of achieving 99.9%+ purity levels whilst maintaining economic viability.
The rare earth industry faces additional complexity from varying ore compositions that require process flexibility to handle different actinide-to-lanthanide ratios across different deposits and processing campaigns. Furthermore, implementing a robust critical minerals strategy becomes essential for managing these diverse feedstock challenges.
Advanced Materials Manufacturing Needs
High-technology applications including permanent magnet production, optical materials, and electronic components require rare earth materials with extremely low actinide contamination levels. Even trace actinide content can compromise performance in precision applications or create regulatory compliance issues for finished products.
Manufacturing sectors driving separation demand include:
• Electric vehicle motor production requiring high-purity neodymium-praseodymium
• Defence electronics manufacturing with strict material specifications
• Renewable energy systems where material longevity depends on chemical purity
• Medical imaging equipment requiring lanthanide materials free from radioactive contamination
What Are the Current Technical Bottlenecks in Scaling These Processes?
Industrial scaling of actinide-lanthanide separation faces multiple technical challenges that significantly impact both capital investment requirements and operational economics. These bottlenecks represent fundamental engineering challenges that require specialised solutions beyond conventional chemical processing approaches.
Equipment Design and Engineering Challenges
Radiation exposure considerations fundamentally alter equipment design requirements for actinide separation facilities. Materials selection must account for radiation-induced degradation, while system accessibility requires remote handling capabilities that increase both complexity and cost significantly compared to conventional rare earth processing.
Corrosion management represents another critical challenge, as the acidic conditions and specialised extractants used in separation processes can rapidly degrade standard metallurgical materials. Equipment must withstand not only chemical attack but also radiation-enhanced corrosion mechanisms that accelerate material degradation rates.
Engineering complexity factors:
• Radiation shielding integration into process equipment
• Remote maintenance access for contaminated systems
• Specialised materials resistant to radiation and chemical attack
• Containment systems for radioactive process streams
Process Optimisation and Automation Requirements
Modern separation processes increasingly rely on automated control systems to maintain the precise operating conditions necessary for high selectivity. According to research on advanced separation technologies, Bayesian optimisation techniques have demonstrated the ability to reduce experimental screening requirements by up to 74% whilst maintaining separation performance. However, implementing these systems requires significant technical expertise.
Traditional manual operation approaches prove inadequate for the tight parameter control required in actinide separation, necessitating investment in advanced process control systems. These automation requirements increase both capital costs and operational complexity compared to conventional rare earth processing facilities.
Regulatory Compliance and Safety Protocols
Regulatory frameworks governing actinide processing impose requirements that significantly exceed those for conventional rare earth operations. Licensing processes can extend for multiple years and require demonstration of technical competence that few organisations possess outside national laboratory systems.
Safety protocols must address both chemical hazards common to rare earth processing and radiological hazards unique to actinide handling. This dual requirement necessitates specialised training programmes and safety equipment that substantially increase operational costs and complexity.
How Are Different Countries Approaching Separation Technology Development?
National approaches to actinide-lanthanide separation technology development reflect broader strategic priorities regarding nuclear fuel cycles, rare earth supply security, and technological independence. These divergent strategies create varying levels of technical capability and industrial capacity across different regions.
United States Research and Development Programmes
The United States focuses primarily on nuclear waste management applications through national laboratory research programmes and Department of Energy funding initiatives. ARPA-E has provided significant support for advanced separation technologies, including electrochemical approaches that could reduce processing complexity and environmental impact.
American companies including Phoenix Tailings and Energy Fuels leverage existing uranium processing expertise to address actinide separation challenges. Energy Fuels, with established uranium processing capabilities, represents one of the most technically capable organisations outside China for handling actinide-contaminated rare earth materials.
US development priorities:
• Advanced nuclear fuel cycle technologies
• Rare earth supply chain diversification
• Electrochemical processing innovation
• Automated separation system development
European Union Strategic Initiatives
European approaches emphasise sustainable processing technologies and regulatory compliance frameworks that could enable broader deployment of separation capabilities. The EU's Critical Raw Materials Act creates policy drivers for developing domestic separation capacity, particularly for materials where China currently maintains dominant market positions.
REEtec in Norway exemplifies European strategy through partnerships with established mining companies like LKAB to create integrated Nordic value chains. French company Carester appears positioned to emerge as a European separation leader through advanced technology development and strategic partnerships. Additionally, establishing a critical raw materials facility remains central to Europe's supply security objectives.
Asian Technology Development Pathways
Japan and South Korea pursue separation technology development primarily to support advanced nuclear fuel cycle programmes rather than rare earth industry applications. Both countries maintain significant technical capabilities through national research institutions and have developed specialised expertise in high-radiation environment processing.
China's dominance in rare earth separation extends to actinide-lanthanide separation capabilities, though specific technical details remain limited due to proprietary considerations and export control restrictions on related technologies.
The next major ASX story will hit our subscribers first
What Role Does Automation Play in Modern Separation Facilities?
Automation has become essential for achieving consistent performance in actinide-lanthanide separation processes, where manual control cannot maintain the precision required for optimal selectivity. Advanced control systems enable operation at parameter tolerances that would be impossible to achieve through conventional manual oversight approaches.
High-Throughput Screening Technologies
Automated screening systems enable rapid evaluation of multiple process conditions simultaneously, accelerating optimisation of separation parameters. These systems can evaluate hundreds of extraction conditions per day compared to dozens achievable through manual laboratory work, dramatically reducing development timelines.
High-throughput approaches prove particularly valuable for actinide separation development, where radiation safety considerations limit manual handling capabilities. Automated systems can operate in high-radiation environments whilst maintaining precise control over experimental parameters.
Screening system capabilities:
• Parallel evaluation of multiple extractant formulations
• Automated pH adjustment and monitoring
• Real-time analytical feedback for process optimisation
• Statistical analysis integration for rapid parameter identification
Bayesian Optimisation for Process Control
Machine learning approaches, particularly Bayesian optimisation, enable intelligent exploration of process parameter space to identify optimal operating conditions with minimal experimental effort. These methods have demonstrated significant efficiency improvements in separation process development and optimisation.
Bayesian methods prove especially valuable for actinide separation optimisation, where the high cost and safety requirements of experimental work make exhaustive parameter screening impractical. The approach can identify near-optimal conditions with 74% fewer experiments than traditional screening approaches.
Remote Handling and Safety Systems
Automation enables separation processing in high-radiation environments where direct human access would be impossible or unsafe. Remote handling systems allow maintenance and operation of separation equipment whilst maintaining radiation exposure within acceptable limits.
Modern facilities integrate data-driven operations and robotic systems for sample handling, equipment maintenance, and waste management operations. These systems require sophisticated control algorithms to maintain separation performance whilst operating under remote control conditions.
Which Specific Extractants Show the Most Promise for Future Applications?
Advanced extractant development focuses on compounds offering improved selectivity, radiation stability, and operational simplicity compared to traditional separation reagents. These next-generation materials could enable broader deployment of separation technologies by addressing current technical and economic limitations.
TODGA and TEHDGA Performance Characteristics
N,N,N',N'-tetraoctyldiglycolamide (TODGA) represents a proven diglycolamide extractant with demonstrated performance in pilot-scale operations. TODGA achieves moderate selectivity factors (2-4) for actinide-lanthanide separations whilst offering enhanced radiation stability compared to phosphorus-based alternatives.
TEHDGA (N,N,N',N'-tetrakis-2-ethylhexyldiglycolamide) demonstrates improved selectivity (factors of 3-6) through optimised alkyl chain structure that enhances metal complex formation selectivity. The compound shows compatibility with multiple acid systems, enabling process flexibility not available with nitric acid-specific extractants.
TODGA advantages:
• Proven performance in pilot operations
• Enhanced radiation tolerance
• Simplified back-extraction chemistry
• Reduced organic phase degradation
DMDODGA Structural Optimisation Benefits
Dimethyl-dioctyl-diglycolamide (DMDODGA) incorporates structural modifications designed to reduce steric hindrance during metal complex formation. These modifications enable improved selectivity whilst maintaining the radiation stability advantages of the diglycolamide extractant family.
The compound shows particular promise for hydrochloric acid systems, expanding processing options beyond nitric acid-based flowsheets. This acid compatibility could enable integration with existing rare earth processing facilities without requiring complete process redesign.
Next-Generation Extractant Development
Research programmes focus on developing extractants that combine high selectivity with simplified process chemistry and reduced environmental impact. Target characteristics include biodegradability, reduced toxicity, and compatibility with green chemistry principles whilst maintaining separation performance.
Emerging extractant classes under development include:
• Functionalised ionic liquids for enhanced selectivity
• Biomimetic extractants based on naturally occurring compounds
• Hybrid extractants combining multiple separation mechanisms
• Solid-supported extractants for simplified processing
| Extractant Class | Selectivity Range | Development Status | Key Innovation |
|---|---|---|---|
| Advanced DGA | 2-8 | Pilot demonstration | Structural optimisation |
| Ionic Liquids | 5-15 | Laboratory research | Tunable properties |
| Biomimetic | 3-12 | Early research | Environmental compatibility |
| Hybrid Systems | 8-25 | Conceptual development | Multiple mechanisms |
How Do Environmental and Safety Considerations Impact Process Design?
Environmental and safety requirements fundamentally shape actinide-lanthanide separation facility design, often driving technology selection and operational approaches more than purely technical performance criteria. These considerations create significant cost implications that must be balanced against separation performance requirements.
Radioactive Material Handling Requirements
Radiation protection protocols require comprehensive shielding, contamination control, and exposure monitoring systems that substantially increase facility complexity and cost. Design must accommodate not only direct radiation from actinide materials but also activated components and contaminated equipment throughout the processing system.
Ventilation systems must maintain negative pressure gradients to prevent radioactive contamination release whilst managing the chemical fumes generated by separation processes. This dual requirement necessitates sophisticated air handling systems with multiple filtration stages and monitoring capabilities.
Safety system requirements:
• Multi-stage HEPA filtration for exhaust systems
• Continuous air monitoring for radioactive and chemical contamination
• Emergency shutdown systems for process isolation
• Decontamination facilities for equipment and personnel
Waste Stream Management and Minimisation
Separation processes generate multiple waste streams containing varying levels of radioactive and chemical contamination. Waste minimisation strategies must address both volume reduction and contamination level management to optimise disposal costs and regulatory compliance.
Secondary waste generation from contaminated equipment, protective clothing, and maintenance materials can exceed primary process waste volumes. Design approaches that minimise secondary waste generation through remote handling and simplified equipment access can significantly reduce overall waste management costs.
Solvent recycling and regeneration systems enable substantial reduction in organic waste volumes whilst recovering expensive extractants for reuse. However, these systems add complexity and require careful monitoring to prevent contamination buildup that could compromise separation performance. Implementing comprehensive waste management solutions becomes crucial for maintaining operational efficiency.
Green Chemistry Approaches in Extractant Development
Environmental sustainability considerations increasingly influence extractant selection and process design decisions. Green chemistry principles promote use of less toxic materials, renewable feedstocks, and biodegradable compounds where technically feasible.
Water-based processing systems receive increasing attention as alternatives to organic solvent-based extraction, though achieving equivalent selectivity remains challenging. Supercritical fluid extraction using carbon dioxide represents another environmentally preferable approach under development for specific applications.
What Are the Economic Factors Driving Separation Technology Investment?
Economic considerations for actinide-lanthanide separation technology investment involve complex trade-offs between capital requirements, operational costs, and market value creation. These factors vary significantly between nuclear waste management applications and rare earth industry applications, creating different investment drivers for each sector.
Capital Equipment and Infrastructure Costs
Separation facility capital costs significantly exceed those for conventional rare earth processing due to radiation handling requirements and specialised equipment needs. Shielding, remote handling systems, and radiation-resistant materials can increase facility costs by 300-500% compared to conventional chemical processing plants.
Equipment procurement faces additional challenges from limited supplier bases with nuclear-qualified manufacturing capabilities. This constraint increases both capital costs and delivery timelines, affecting project economics through extended development schedules and higher financing costs.
Major capital cost components:
• Radiation shielding and containment structures
• Remote handling and automation systems
• Specialised materials and fabrication requirements
• Regulatory compliance and licensing costs
Operating Expense Considerations
Operational costs include specialised labour requirements, enhanced safety protocols, and waste management expenses that exceed conventional processing operations. Maintenance costs increase substantially due to radiation exposure limitations and requirements for specialised procedures and equipment.
Extractant costs represent a significant operational expense, particularly for advanced reagents with limited commercial availability. Solvent losses and degradation rates in high-radiation environments can substantially exceed those in conventional processing, increasing reagent replacement costs.
Return on Investment Calculations for Industrial Applications
Investment returns vary dramatically between applications, with nuclear waste management typically justified through regulatory compliance requirements rather than direct economic returns. Rare earth applications must demonstrate positive economics through premium pricing for high-purity products or access to previously unusable feedstocks.
Market pricing for separation services varies significantly based on feed material characteristics, required purity levels, and throughput requirements. Premium pricing for actinide-free rare earth products can justify investment in separation capabilities for suppliers serving regulated markets.
Which Emerging Technologies Could Transform Future Separation Processes?
Transformational separation technologies under development could dramatically alter the economics and technical feasibility of actinide-lanthanide separation. These approaches address fundamental limitations of current methods whilst potentially enabling broader deployment of separation capabilities across different industry sectors.
Advanced Materials for Selective Adsorption
Engineered adsorbent materials offer potential alternatives to liquid-liquid extraction through selective binding mechanisms designed specifically for actinide-lanthanide discrimination. These materials could enable simplified processing flowsheets with reduced chemical inventory and waste generation.
Functionalised nanoparticles demonstrate enhanced selectivity through precisely controlled surface chemistry and binding site architecture. Metal-organic frameworks (MOFs) and similar structured materials enable tunable selectivity through systematic modification of pore size and surface functionality.
Advanced adsorbent advantages:
• Simplified process flowsheets compared to liquid extraction
• Reduced chemical inventory requirements
• Enhanced selectivity through engineered binding sites
• Potential for continuous processing configurations
Microfluidic and Continuous Processing Systems
Microfluidic separation systems enable precise control of extraction conditions whilst reducing equipment footprint and chemical inventory. These systems demonstrate enhanced mass transfer rates through increased surface-to-volume ratios, potentially enabling faster separation kinetics and reduced residence times.
Continuous processing approaches could eliminate batch-to-batch variability whilst enabling better integration with automated control systems. The reduced system volume inherent in microfluidic approaches offers safety advantages through minimised radioactive material inventory.
Artificial Intelligence Integration in Process Control
Machine learning algorithms enable real-time optimisation of separation parameters based on continuous monitoring of process conditions and product quality. These systems can adapt to feedstock variations and equipment performance changes without manual intervention.
Predictive maintenance capabilities using AI analysis of equipment performance data could substantially reduce maintenance costs and unplanned downtime in high-radiation environments where equipment access is limited and expensive.
AI integration benefits:
• Real-time process optimisation
• Predictive equipment maintenance
• Automatic adaptation to feedstock variations
• Enhanced safety through intelligent monitoring
Future Outlook for Actinide-Lanthanide Separation Technology
The trajectory of actinide-lanthanide separation technology development reflects converging drivers from nuclear waste management requirements, rare earth supply security concerns, and advancing automation capabilities. These factors suggest accelerating investment and deployment over the next decade.
Projected Technology Developments Through 2030
Technical advancement priorities focus on reducing operational complexity whilst maintaining separation performance. Automated systems incorporating machine learning optimisation could enable broader deployment by reducing the specialised expertise currently required for successful operations.
Advanced extractants nearing commercial deployment offer improved selectivity and simplified process chemistry compared to current methods. These developments could enable separation facility operation by organisations without extensive nuclear processing experience.
Expected developments include:
• Commercial deployment of advanced DGA extractants
• Integration of AI-based process control systems
• Demonstration of microfluidic separation technologies
• Enhanced radiation-tolerant automation systems
Market Demand Drivers and Growth Projections
Market growth drivers include expanding nuclear power deployment, increasing rare earth demand from clean energy technologies, and policy initiatives promoting supply chain diversification. These factors collectively support substantial investment in separation capability development.
Regulatory requirements for radioactive waste management create sustained demand for separation technologies regardless of economic cycles. This demand provides a stable foundation for technology development and deployment investment.
Strategic Implications for Global Supply Chains
Separation technology deployment outside China could fundamentally alter rare earth supply chain dynamics by enabling processing of previously unusable ore deposits containing actinide contamination. This capability expansion could reduce dependence on Chinese separation capacity whilst accessing new raw material sources.
Nuclear fuel cycle applications for separation technology may drive broader technology development that benefits rare earth industry applications through shared technical advances and reduced development costs.
"Distribution ratios for americium extraction typically exceed 1.0 across nitric acid concentration ranges, indicating preferential organic phase partitioning. Modern analytical approaches using Bayesian optimisation have reduced experimental screening requirements by 74% whilst maintaining separation efficiency standards."
The challenge of separating actinides from rare earths represents one of the most technically demanding separations in modern chemistry. As industry advances toward more sophisticated processing technologies and environmental requirements become increasingly stringent, the successful development and deployment of these separation technologies will prove crucial for both nuclear waste management and critical mineral supply chains.
Disclaimer: This analysis presents current technical understanding and market projections based on available information through 2024. Actual technology development timelines, commercial deployment success, and market dynamics may vary significantly from projections due to technical, regulatory, and economic factors beyond current prediction capabilities. Investment decisions should consider substantial technical and market risks associated with emerging separation technologies.
Looking to Capitalise on Critical Minerals Processing Opportunities?
Discovery Alert's proprietary Discovery IQ model delivers instant notifications on significant ASX mineral discoveries, including breakthrough advances in rare earth processing and separation technologies that could transform entire supply chains. Explore how major mineral discoveries have generated exceptional returns by examining Discovery Alert's comprehensive database of historic market-moving announcements. Begin your 30-day free trial today to position yourself ahead of critical minerals developments that could reshape industrial processing capabilities.